19 August 2022
The risk of developing a subtype of leukaemia known as iAMP21-ALL is amplified in individuals that carry the translocated chromosome rob(15;21)c. Connor Gilkes-Imeson used a Travelling Fellowship from Journal of Cell Science to learn 3D correlative light electron microscopy (3D-CLEM), a technique that he plans to apply in his studies of the kinetochore attachments that form at this mutated chromosome.
Translocated chromosome rob(15;21)c has two centromeres instead of the usual one, and is produced via the breakage and subsequent joining together of chromosomes 15 and 21. Centromeres are important for proper cell division, as they are where the kinetochores assemble in preparation for attachment to the spindle microtubules. Once properly attached, these microtubules begin to shorten, pulling the sister chromatids to opposite poles of the cell. Connor and his colleagues found that rob(15;21)c is more likely to mis-segregate during mitosis than healthy chromosomes, and he wanted to determine whether this is due to abnormal attachments between its kinetochores and the spindle microtubules.
Connor used a Travelling Fellowship to visit Dr Daniel Booth’s laboratory at the University of Nottingham, where he spent three weeks learning how to perform 3D-CLEM. This challenging technique involves preparing a single sample so that it can be imaged using both light and electron microscopy. Connor’s time in the Booth lab allowed him to follow the whole process, from sample preparation and imaging on the University of Nottingham’s focused ion beam scanning electron microscope (FIB-SEM), to creating 3D models from the data.
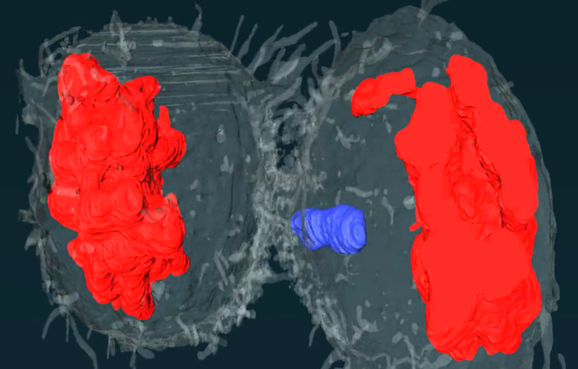
During his Travelling Fellowship, Connor was able to produce images showing dividing cells with lagging chromosomes. These preliminary data demonstrate the potential power of the 3D-CLEM technique, and Connor hopes to optimise his pipeline to improve the image resolution now that he is back at his home lab in Newcastle, where he is jointly supervised by Professor Jonathan Higgins and Professor Christine Harrison. 3D-CLEM is not widely used at Newcastle University, so Connor also hopes to be able to advise other researchers who might want to try the approach.
“The Travelling Fellowship from The Company of Biologists gave me the opportunity to travel to Nottingham University and learn new laboratory techniques,” Connor said. “I will now be able to use these techniques at my home lab in Newcastle and they will be extremely valuable for my research. I would undoubtedly recommend the Travelling Fellowship to anybody that is considering applying.”


You must be logged in to post a comment.