Credit: Ernesto del Aguila III, National Human Genome Research Institute, NIH
Targeted mutagenesis in a human-parasitic nematode
Spencer S. Gang, Michelle L. Castelletto, Astra S. Bryant and Elissa A. Hallem
Department of Microbiology, Immunology, and Molecular Genetics, University of California, Los Angeles, USA
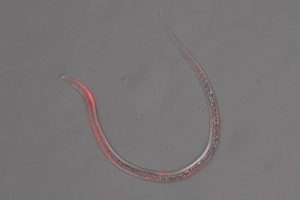
Human-parasitic nematodes cause extensive morbidity worldwide, particularly in low-resource settings. Despite their prevalence, our understanding of the biology of parasitic nematodes has been limited by the lack of tools for genetic intervention. In particular, it was not possible to generate targeted gene disruptions and mutant phenotypes in any parasitic nematode. We recently developed a method for introducing CRISPR/Cas9-mediated gene disruptions in the human-parasitic skin-penetrating nematode Strongyloides stercoralis. As a proof-of-concept, we first disrupted the S. stercoralis twitchin gene Ss-unc-22. Disruption of Ss-unc-22 resulted in nematodes with severe motility defects. Ss-unc-22 mutations were resolved by homology‑directed repair when a repair template was provided. Omission of a repair template resulted in deletions at the target locus. Ss-unc-22 mutations were heritable; we passed Ss‑unc‑22 mutants through a host and successfully recovered mutant progeny. Using a similar approach, we also disrupted the unc-22 gene of the rat-parasitic nematode Strongyloides ratti. These results demonstrated the applicability of CRISPR/Cas9 to parasitic nematodes. We then used this system to identify genes required for host seeking in S. stercoralis. In C. elegans, many sensory behaviors require the cGMP-gated cation channel subunit gene tax-4. We found that targeted mutagenesis of the S. stercoralis tax-4 homolog abolishes sensory-driven host seeking, demonstrating that parasitic host-seeking behaviors are generated through an adaptation of sensory cascades that drive environmental navigation in C. elegans. Our results provide insight into the molecular mechanisms by which human-parasitic nematodes find and infect human hosts, and enable future studies of gene function in these medically relevant parasites.


You must be logged in to post a comment.