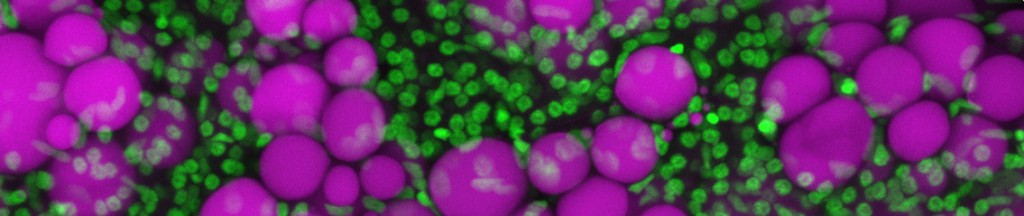
Photo credit: James Minchin (University of Edinburgh) and John Rawls (Duke University)
Non-adrenergic control of fat storage and metabolism in adipose tissues
Martin Klingenspor
School of Life Sciences, Technische Universität München, Germany
Adipocytes are highly flexible in their capacity to store and mobilize fatty acids according to local and systemic energy demands. In mammals, the metabolic phenotype of adipocytes ranges from fat-laden unilocular white adipocytes with slow energy turnover to multilocular brite (or beige) and brown adipocytes equipped for nonshivering thermogenesis. Beyond these opposing functions in energy storage, mobilization and thermogenesis, adipocytes also secrete adipokines known to affect cellularity, metabolic capacities of adipocytes, inflammation, and target neuroendocrine control of energy balance in the brain and systemic metabolism. Differential adipokine profiles of white, brite and brown adipocytes may reflect their metabolic functions. Moreover, adipose tissues harbor macrophages and T-cells involved in tissue homeostasis and adipose-specific immune defense. These processes are subject to neuronal control by the sympathetic innervation of adipose tissues. During the past years, however, a number of non-adrenergic effectors have been identified to modulate adipose tissue heterogeneity and function. To sort out this complexity, several questions need to be addressed:
- Are the published biological effects robust and reproducible?
- Are non-adrenergic effectors acting independent of the sympathetic innervation?
- Do their cellular signaling pathways cross-talk with the pathways triggered by adrenergic receptors?
- Do these effectors trigger acute metabolic responses or long-term adaptation of tissue functions?
- Are we dealing with general mechanisms or specific adaptations on the species level?
Comparative aspects of adipose tissue biology may reveal valuable information in this context.


You must be logged in to post a comment.